Posted by
Jenny Garner
14 Comments
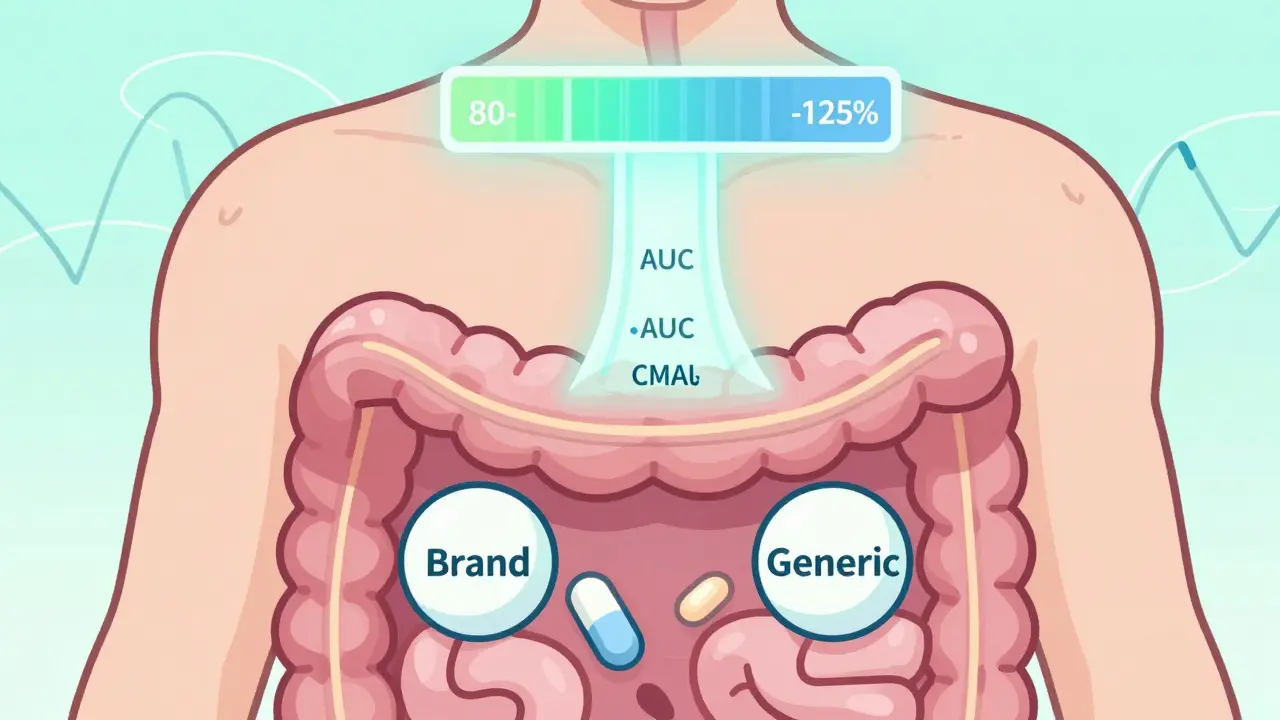
The 80-125% rule ensures generic drugs perform like brand-name versions by testing how much drug enters your bloodstream. It's not about pill strength - it's about absorption. Learn how regulators use pharmacokinetics to guarantee safety and effectiveness.
read morePosted by
Paul Fletcher
10 Comments

Generic drugs are just as safe and effective as brand-name medications but cost up to 85% less. Learn how they’re made, tested, and approved by the FDA through the ANDA process-step by step.
read more